IFNα release assay
Interferon alpha (IFNα) is a main driver of systemic autoimmune diseases such as SLE and myositis, and its overexpression in patients with these diseases is in part caused by autoantibody-containing immune complexes. Immune complexes containing autoantibodies bound to nucleic acids can stimulate innate cell types, such as plasmacytoid dendritic cells (pDC), to secrete IFNα. By binding to cell surface receptors for immune complexes (FcR), immune complexes containing in particular RNA are endocytosed and stimulate IFNα production by interacting with toll like receptors (TLR), such as TLR7.
To investigate whether chemical inhibitory probes and tool compounds can affect INFα secretion induced by autoantibody-containing immune complexes.
Chemical probes MK-2206 and skepinone inhibiting kinases AKT and p38-MAPK, respectively, reduce INFα secretion. In addition, the chemical probe UNC0638 targeting the methyltransferase G9a and likely unidentified off-target proteins, reduces IFNα secretion.
iconIFNα assay dataset
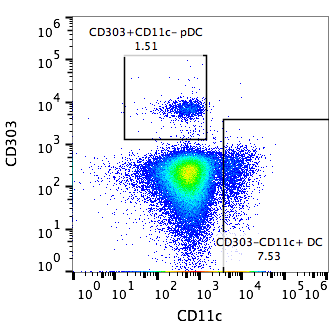
General protocol: PBMC (Peripheral Blood Mononuclear Cells) are cultured for 24 hours in presence of SLE-derived immune complexes. The effect on IFNα secretion by addition of chemical probes and tool compounds is investigated by ELISA. The frequency of pDC in the PBMC preparation is investigated by flow cytometry.
Cell culture condition: Peripheral blood mononuclear cells (PBMC) are cultured at 2,5 million/ml, 200µl/well, in triplicates. Culture medium is RPMI supplemented with 10% heat inactivated bovine serum. Cells are pre-incubated with probes and chemical tool compounds (1 μM or 0.1 μM) 30 min before addition of SLE-derived immune complexes (anti-RNP+ IgG from SLE sera 100mg/ml mixed with apoptotic celline U937, kind gift from Pfizer).
Readout: Flow cytometric analysis is performed on freshly isolated PBMC, and include staining of CD303, CD11c, CD14, CD3 and a marker for cell viability. Cell culture supernatants are removed after 24 hours and analysed for IFNα using a pan-IFNα specific ELISA. Controls include unstimulated condition (cell culture medium without added immune complexes), stimulated condition with vehicle control only (DMSO), stimulated condition with IRAK 1/4 inhibitor I.
We investigated INFα release upon 24 hours stimulation with SLE immune complex in 30 blood donors including 15 patients with systemic lupus erythemtosus (SLE), 8 with idiopathic inflammatory myositis (IIM) and 7 healthy donors. SLE patients were subdivided into hydroxychloroquine-treated and -untreated patients, and IIM patients into those with dermatomyositis (DM) and polymyositis (PM). Immune complex induced variable levels of IFNα with lower levels in SLE patients compared to healthy donors (p<0,05). Of note, 6 of the 10 SLE patients treated with hydroxychloroquine (HCQ) presented with low IFNα induction (<100 pg/ml) and were removed from further analyses (U17, U27, U37, U39, U40, U41).
Fig 1
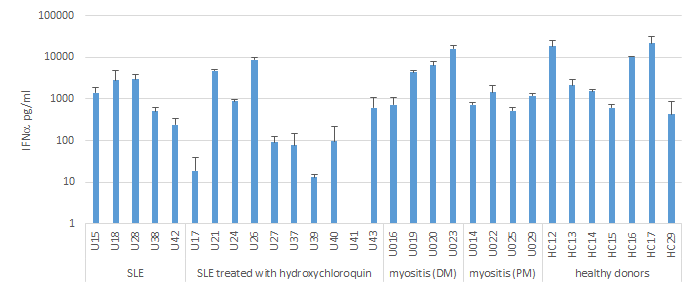
IFNα released after 24h stimulation of PBMC with SLE immune complex. Data are depicted from 30 individual blood donors, as average+SD of biological triplicates.
Although pDC are considered the most efficient producers of IFNα in blood, the frequency of pDC in the cell culture was not found to correlate to immune complex induced IFNα levels. There was no difference in frequency of pDC between groups of blood donors. The six SLE patients treated with hydroxychlorine and with low IFNα induction did not differ in the frequency of pDC to the rest of the blood donors (Fig 2b, red dots).
Fig 2
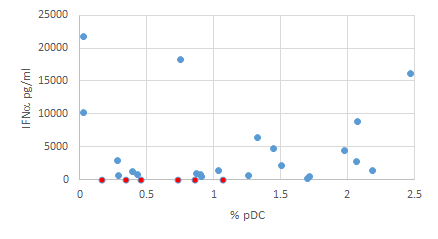
IFNα released after 24h stimulation of PBMC with SLE immune complex vs the percent of pDC of CD3-CD14- live PBMC. Data are depicted from 30 individual blood donors. SLE patients treated with hydroxychloroquin and with <100 pg/ml IFNα induction are marked in red.
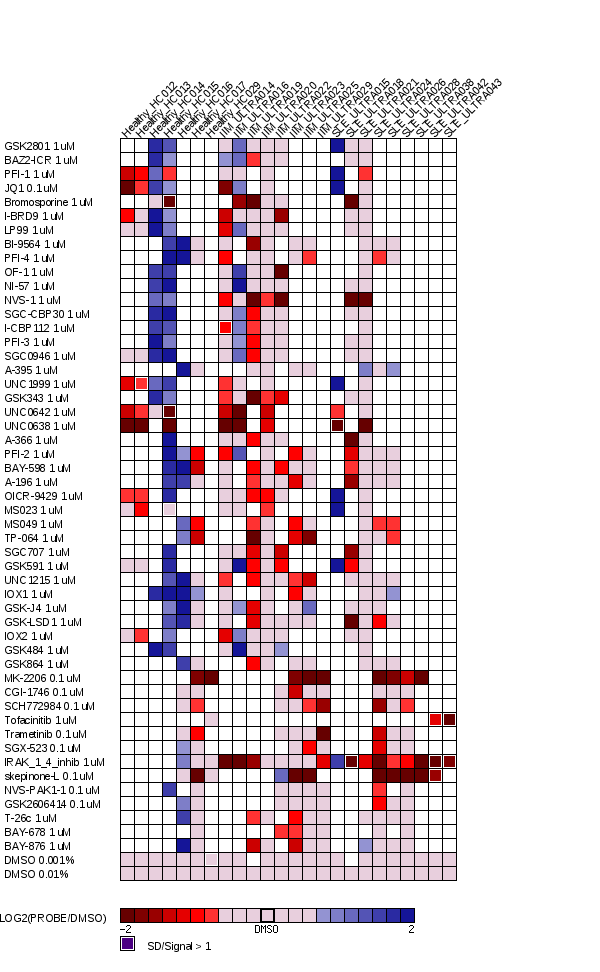
A set of 51 probes were tested in 24 blood donors including 9 patients with systemic lupus erythemtosus (SLE), 8 with idiopathic inflammatory myositis (IIM) and 7 healthy donors. The effect on PBMC IFNα secretion after 24h cell culture in presence of SLE immune complex is expressed normalized to the vehicle control (0,01% or 0,001% DMSO). Probes are added at 1uM or 0,1uM, as indicated. Pobes with p<0,07 vs vehicle control are marked with red boxes. Due to constraints in the number of PBMC available from each donor, not all probes were tested in each blood donor.
Fig 3
The results from individual donors of the 9 probes with p<0,07 vs vehicle control is shown in figure 4. Of these, UNC0638, MK-2206 and skepinone showed significant inhibitory effects (p<0,05 and >50% average inhibition). The effects of the different probes did not differ between groups of blood donor. Thus, we confirm AKT and p38-MAPK as important players in immune complex induced cellular activation. The inhibitory effect of UNC0638 may indicate off target interactions, which are presently being investigated. We saw variable but significant inhibitory effect using the IRAK 1/4 inhibitor I at 1uM.
Fig 4